🌟 Improve your sports field with a expert audit.
🌟 Improve your sports field with a expert audit.

Share on RRSS

Water in water systems irrigation may cause corrosion and scaling depending on its composition.
The water There is no such thing as pure water. All water contains dissolved salts. All chemical elements present in an irrigation water are present in the form of different chemical species.
For example, calcium can be present in water as calcium ion. As part of the Calcium Sulphate (Gypsum) molecule, and in turn also as part of the Calcium Carbonate molecule (Calcite).
In certain situations, e.g. due to temperature rises (summer season), any of these chemical species may no longer be dissolved.
It is at this point that we say that it is supersaturated in a given species, its saturation index is greater than 0.5, IS > 0.5, and it can begin to "...".precipitate“.
Salts decrease the effective diameters in pipes by concentric calcareous deposition, with a decrease in pipe diameter and pressure rating.
All this leads to a decrease in the efficiency of the hydraulic system of the field and the appearance of "...".dry patches"This is a possible consequence of insufficient irrigation.
The opposite is the case where the irrigation water, due to its very nature, is avid of certain chemical species, and therefore dissolves minerals, even the very ones from which the irrigation pipes were made.
We say that the water is highly unsaturated with these chemical species, its saturation index, IS <0.5, and the "saturation index" occurs.corrosion“.
The ideal situation is one of equilibrium, where neither one phenomenon nor the other occurs. The saturation index for the chemical species under study is between -0.5 and 0.5, and water is said to be in equilibrium.
It is therefore possible to correct these phenomena, either through acid injections or other mechanisms, depending on the particular situation.
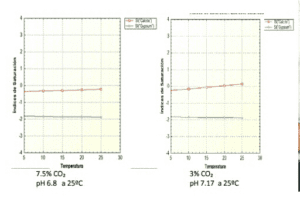
The attached graph shows how these phenomena are produced due to changes in the temperature of the irrigation water or situations of high CO2 content in the soil resulting from the degradation of organic matter in the soil. acid soils.

We appreciate your interest in us, so we leave you this form so that you can subscribe and have priority access to our exclusive promotions and offersideal for saving money on your purchases and keeping your sports and agricultural fields at the forefront!
In addition, we will keep you informed about the LATEST NEWS in Greens and Agriculture with the latest entries from our Greenkeepedia and Agrikipediaincluding innovations, events and interviews with experts.
Click to subscribe now and get exclusive access!