🌟 Improve your sports field with a expert audit.
🌟 Improve your sports field with a expert audit.

Share on RRSS

What is behind the numbers that Big Data gives us? It is interesting to give a greater meaning to certain numerical values that we obtain from our agronomic management. Thus, whenever we look at the numerical value of the parameter pH we think about whether our soil or irrigation water will be basic or acidic. Make no mistake, there is much more information in those numbers.
In other words, at the same soil conditions, a lower pH would mean higher respiration, and as the pH increases, respiration will decrease and, therefore, CO2 will be converted to bicarbonate and ultimately to carbonate, according to the attached graph.

It is also important to bear in mind, especially on sand-based sports surfaces, that this partial pressure of CO2 can be very high, reaching up to 3-4% compared with that present in other soils or with the atmospheric 0.3%. Therefore, when we take a sample to the laboratory and this is extracted from the soil, the sample is degassed and does not take into account the CO2 it had at source, so we can say that the laboratory reading is in error.
If we fertilise with acid reaction fertilisers such as ammonium sulphate, we can know when this nitrogen has been used up in the soil. The value of the Soil pH will fall according to the amount of nitrogen supplied and will rise again to its normal level once it is depleted in the soil. See in the example the decrease of half a pH point as a consequence of the acidity of the ammonium sulphate.

Other very interesting numerical values to evaluate are those comparing irrigation waters with the pore waters of their soil profiles. In other words, the more the values are chemically distant from each other, it means that the irrigation water means little modification in the soil, as in the case of clay soils with high cation exchange capacities, but if they are very close, as in the case of the sandy substrates of greens or football pitches, it means that any change in the irrigation water will mean a change in the soil profile. It is therefore very practical to treat irrigation water to modify the profile of the green. This is very visual through the piper diagrams.
On the other hand, current assumptions such as the calcium carbonate crust accumulation - magnesium in USGA greens profiles and the associated infiltration problems due to irrigation with very alkaline and hard water, are not so obvious on our greens or football pitches (Carrow et al., 1999; Ellis, 2009; Fidanza, 2006; Harivandi, 1999; Simmons, 2010).
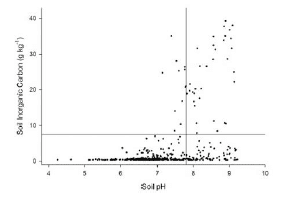
Obear and Soldat (2015) studied the vertical distribution of inorganic carbon in 28 USGA greens and did not observe the accumulations of these carbonates that do occur in agricultural soils, especially in samples with pH below 7.8.
Another value of agronomic interest is the value of the redox of irrigation water. In irrigation water disinfection, monitoring of the redox potential, i.e. the oxidation-reduction potential (ORP), should be required. Several investigations have shown that an ORP value of 650 to 700 mV, bacteria such as Escherichia coli are exterminated within a few seconds. Yeasts and the most sensitive type of fungus spore-forming bacteria are also exterminated at this ORP level after only a few minutes of contact. In 1971, the WHO adopted the REDOX potential as the most reliable measure of water quality.
Make better decisions with us, our top quality services and our more than 15 years of experience in Football and Golf are our guarantee:

We appreciate your interest in us, so we leave you this form so that you can subscribe and have priority access to our exclusive promotions and offersideal for saving money on your purchases and keeping your sports and agricultural fields at the forefront!
In addition, we will keep you informed about the LATEST NEWS in Greens and Agriculture with the latest entries from our Greenkeepedia and Agrikipediaincluding innovations, events and interviews with experts.
Click to subscribe now and get exclusive access!