🌟 Improve your sports field with a expert audit.
🌟 Improve your sports field with a expert audit.

Share on RRSS

The modelling of any system consists of establishing a conceptual model of behaviour that can be replicated, so that by varying the parameters that affect these systems, the results can be predicted before actual changes are made.
For example, it is possible to model the changes that occur in an irrigation water when changing its pH, hydraulic system pressure or temperature, in order, for example, to know the possibility of a water producing calcareous incrustations in the hydraulic systems of the golf course and act accordingly.

When species Calcite or Aragonite (calcium carbonate with different three-dimensional structures) are supersaturated in water, i.e. their saturation indexes are higher than 0.5, they are susceptible to precipitate and cause clogging in sprinklers, etc., although Aragonite does not usually give rise to precipitate nuclei for this purpose. It is therefore preferable for calcareous forms to be in the form of Aragonite rather than Calcite.
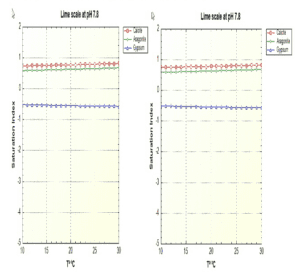
Thus we can foresee how a water with pH 7.8 removes limescale by lowering its pH to 6.5, but does not affect the precipitation of gypsum from the water.

Similarly, gypsum (calcium sulphate) can precipitate if its saturation index is higher than 0.5, which occurs less frequently than in the case of carbonates.
Tiloom offers you the most precise and individual advice on your irrigation water management.

We appreciate your interest in us, so we leave you this form so that you can subscribe and have priority access to our exclusive promotions and offersideal for saving money on your purchases and keeping your sports and agricultural fields at the forefront!
In addition, we will keep you informed about the LATEST NEWS in Greens and Agriculture with the latest entries from our Greenkeepedia and Agrikipediaincluding innovations, events and interviews with experts.
Click to subscribe now and get exclusive access!