🌟 Improve your sports field with a expert audit.
🌟 Improve your sports field with a expert audit.

Share on RRSS

Chlorine is a very frequently used element to sanitise water, but the impact of pH on water disinfection with chlorine is very important.
Chlorine products are widely used to ensure pathogen-free water. These products dissolve in water as hypochlorous acid (HClO). Hypochlorous acid is weak so the dissociation of its hypochlorite ion (ClO-) is very important. As a result, hypochlorite has a very low disinfecting power.
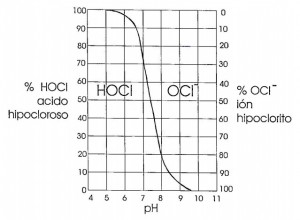
The balance between hypochlorous acid (high disinfectant value) and hypochlorite ion (low disinfectant value) is highly dependent on the pH. As can be seen in the graph, from pH 6 onwards, the disinfecting power drops rapidly.
Before deciding which disinfection system to use it is important to know the nature of the water, write to us so that we can help you design the best disinfection system for your installation.

We appreciate your interest in us, so we leave you this form so that you can subscribe and have priority access to our exclusive promotions and offersideal for saving money on your purchases and keeping your sports and agricultural fields at the forefront!
In addition, we will keep you informed about the LATEST NEWS in Greens and Agriculture with the latest entries from our Greenkeepedia and Agrikipediaincluding innovations, events and interviews with experts.
Click to subscribe now and get exclusive access!